Biocompatibility, low immunogenicity and non-toxicity.
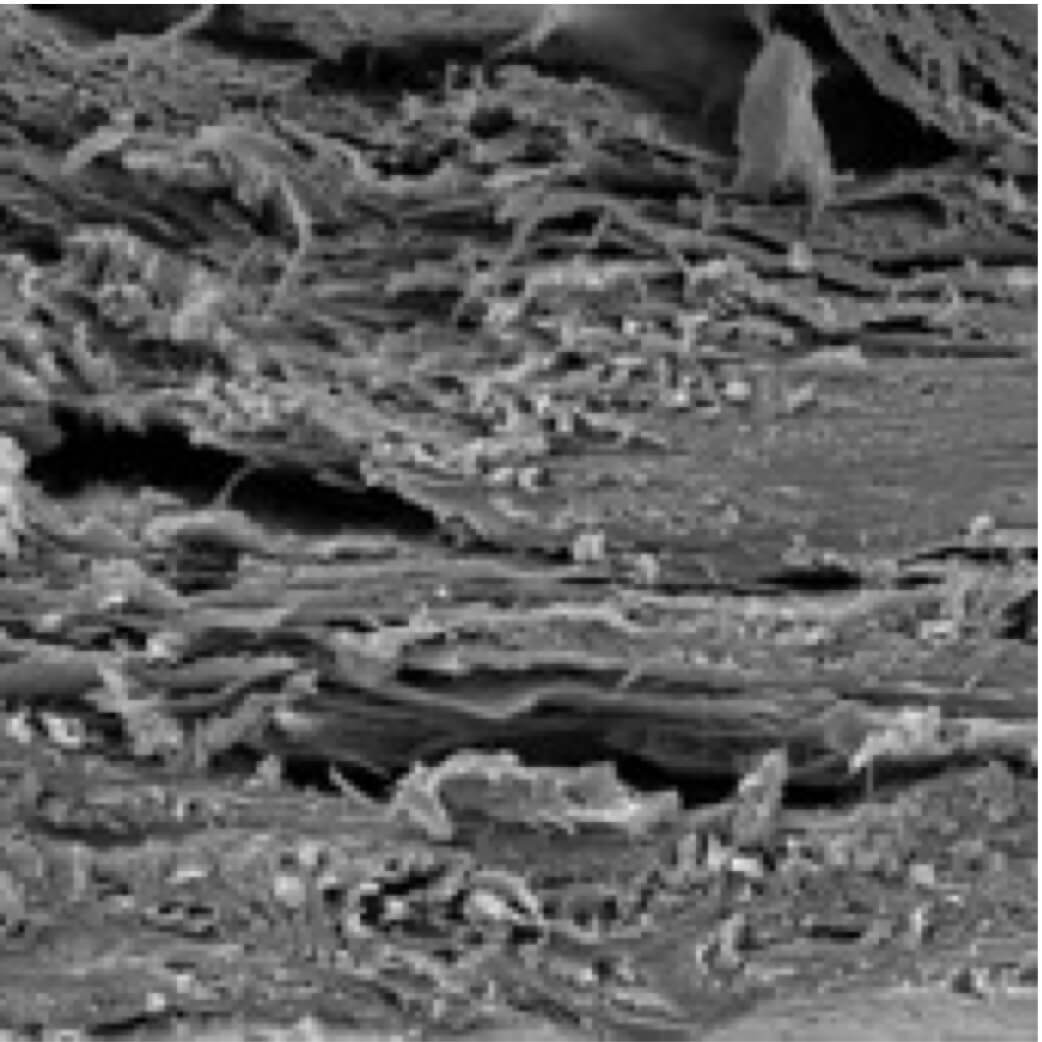
Viscoll® collagen extracted from healthy animal tissues is used to manufacture the Product. Obtaining the extract as a true solution (not a suspension) of native (not denatured) protein provides complete biochemical purification of collagen from potentially immunogenic impurities, which is controlled by appropriate immunological testing. Viscoll® collagen preparations are obtained sterile, apirogenic, completely bioresorbable and irritation-free at implantation, neither in the short nor in the long term.
Adhesive to biological tissues
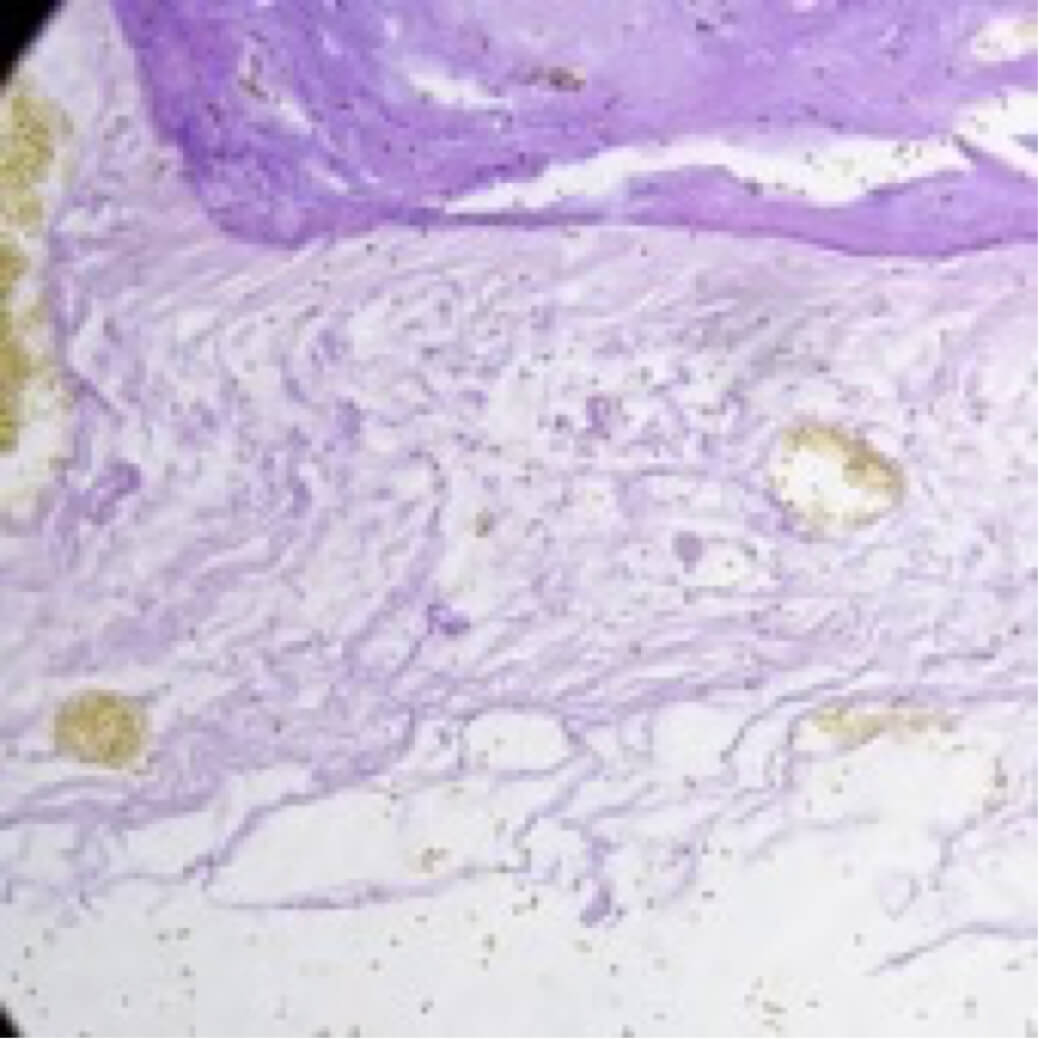
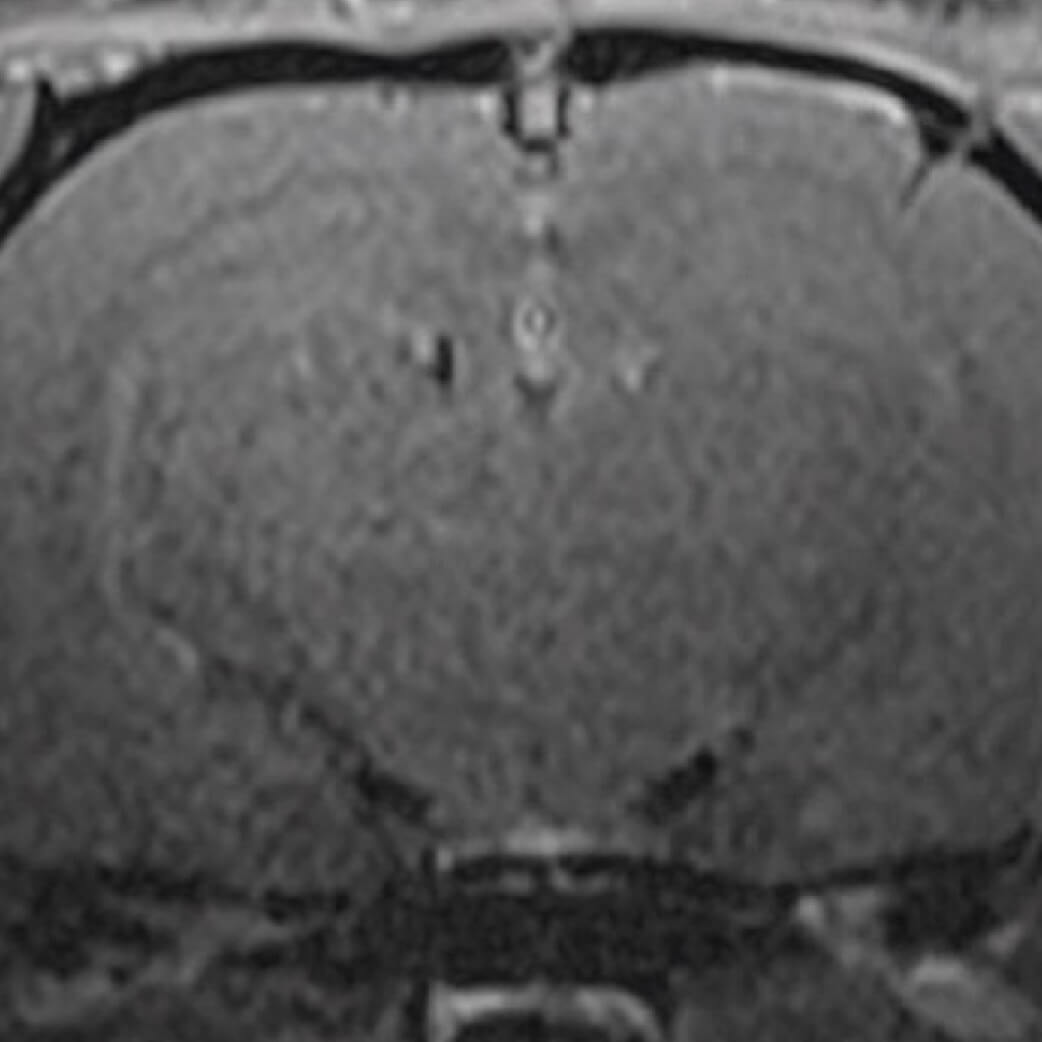
Clear retention of the membrane after application to the affected dura following animal experiments, and prevents the development of cerebrospinal fluid.
Permeable to cells
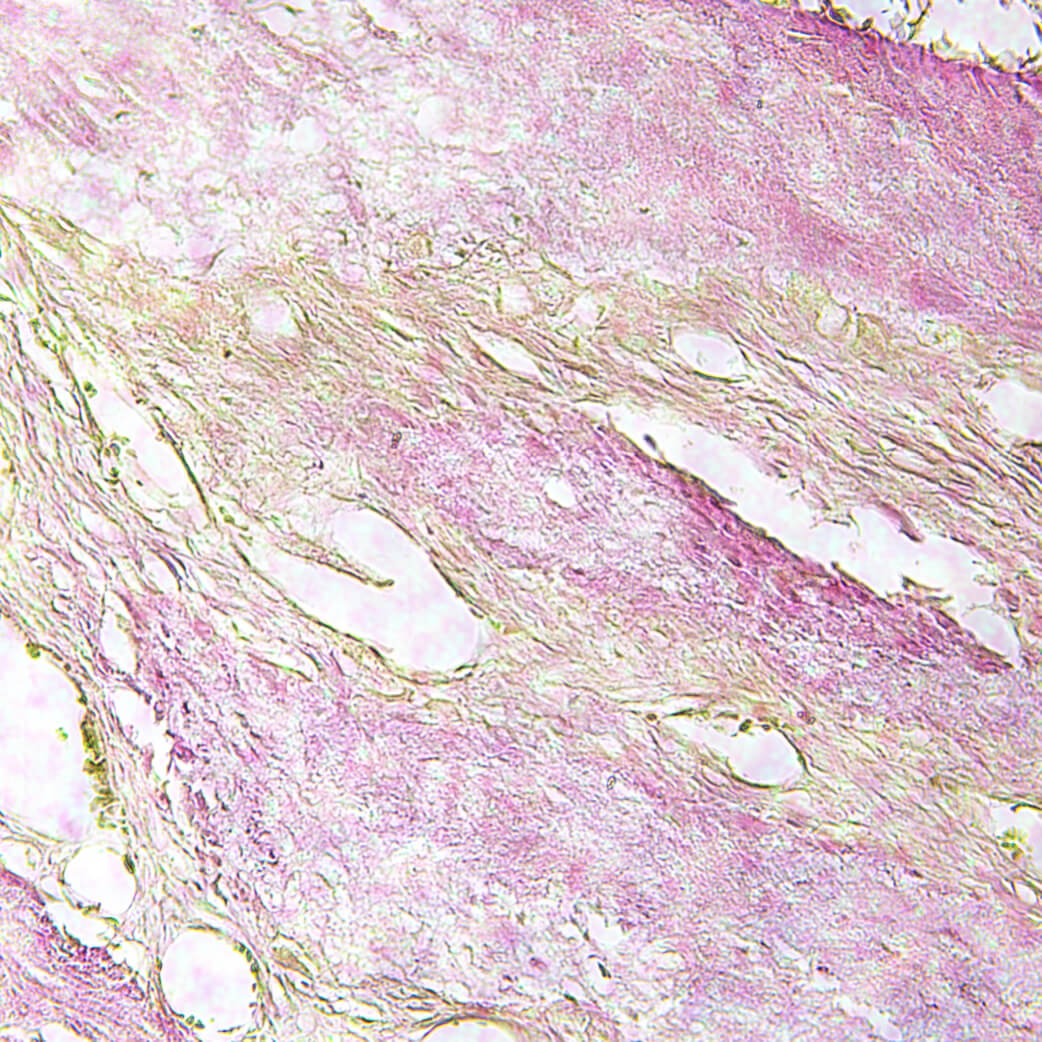
Membrane is permeable to cells both in vitro and in vivo.
Adapted for the cells transplantation
Human cell culture can be pre-cultured on both surfaces of the product.